Covid vaccine winners focus on the flu as their main target

[ad_1]
After convincing the manufacturers of vaccines to make the first shot of Covid-19, Moderna and BioNTech, and their partner Pfizer, are rushing to get them out of another major market: the flu.
They bet that it’s new messenger messenger RNA used on their Covid vaccines for the first time, will allow flu jabs to be quickly developed to meet the ever-changing challenges and improve performance between 40 and 70 percent.
Opponents also force rulers such as Sanofi, Seqirus and GlaxoSmithKline, to speed up their research into other mRNA methods, although they argue that pre-existing materials may be superior.
They have a lot to lose if they go back. According to a study by the Fortune Business Insights group, this large market is expected to grow 7.2 percent annually to $ 10.7bn in 2028. Sanofi predicts that it could be huge, hitting $ 17bn by 2030.
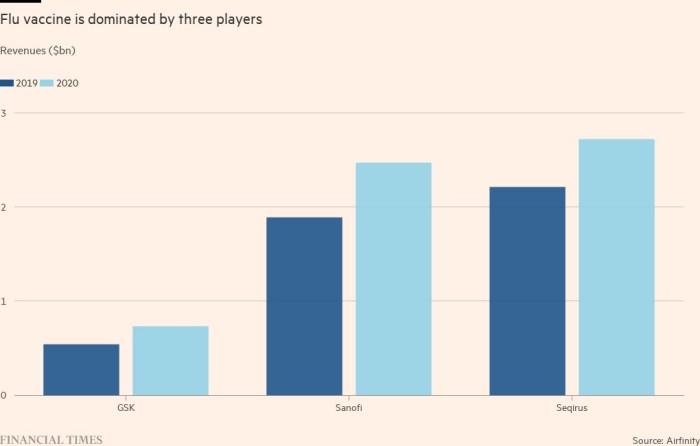
As people became aware of the dangers of respiratory viruses – as well as the potential for vaccines to protect them – funding for the top three influenza vaccine manufacturers increased by 23 to 35 percent from 2019 to 2020. About 40 percent Sanofi € 6bn in annual vaccination costs last came from the flu.
Moderna rocked the competition last week when she announced the first showing that the flu vaccine had increased the number of antibodies in phase 1 trials, even for participants who received the lowest dose. Pfizer and BioNTech began enrolling participants in their Phase 1 study in September.
Stephen Hoge, President of Moderna, told fundraisers that vaccine seekers, who developed an immune system similar to Sanofi market leader in the US Fluzone, were “at the forefront”.
He is a “big believer” in the good that can be combined with the flu vaccine and Covid in a single shot. “We want to be as good as we can get with the extra vaccine,” he said. “And what we want to do is integrate.”
“If [mRNA] it works, you can leave the whole market in Moderna or Pfizer, “said Geoffrey Porges, a reviewer at SVB Leerink Health Bank.
Each year, the World Health Organization (WHO) must predict which strains of the flu will spread next winter. With the modern flu vaccine, they make this choice in early February.
But the production of mRNA, which involves placing the genetic code of the virus in large amounts, is much faster than the old technologies that develop compounds that work in tanks.
Hoge told the Financial Times that this could allow the new winter vaccine to be developed as late as the summer, making it “not just a student comparison”, as there would be more problems circulating in the southern hemisphere.
He said the potential opportunities meant that Moderna’s potential in the flu market was also important as a boost to Covid-19 in the aftermath of the epidemic.
“Right now the flu efficacy prices are bad,” he added.
Subsequently, mRNA can be used to combine vaccines against the four influenza strains set by the WHO, the latest version of Sars CoV-2 and syncytial virus, which can be dangerous for infants and the elderly. Moderna has already been shown to be able to carry six mRNA mutations at a single dose, in a test cytomegalovirus vaccine, which can cause complications in infants and young children.
“We think people don’t need three extras. One pot safe injection sounds like the right answer,” Hoge said.
Mutations in mRNA may also mean that the vaccine may eventually be adapted to the effects of the common cold, or it may be used to make various local vaccines.
Mikael Dolsten, Pfizer’s chief technology officer, told investors that the U.S. pharmacist was “very interested in using mRNA flu” because it could respond better to T cells, white blood cells that are part of the long-term immune system. , changes to modern vaccines.
CureVac, a German specialist who has not yet received the official Covid vaccine, is also hoping to enter the flu market, with fellow UK drugmaker GlaxoSmithKline.
Franz-Werner Haas, CureVac’s chief executive officer, said that artificial intelligence could help predict this kind of future.
GSK did not have much money in its flu business before the outbreak because it adopted traditional methods of breeding. Now, Roger Connor, president of GSK vaccine, said the flu was a “sweet” mRNA, where modern vaccines have so little potential that it is “a rare deficiency”.
“MRNA is something we are investing heavily in right now. We want to be a leader,” he said.
CureVac and GSK have not yet tested clinical trials but Haas said that because Covid vaccine manufacturers are at the forefront, it does not mean they will have the first approved anti-influenza vaccine.
“It’s a competition and we hope everyone benefits,” he said.
But even though Moderna’s phase 1 study showed that its vaccine was safe and produced a strong antibody response, which gave rise to confidence over the next phase of testing, some investors were disappointed.
In another study, the vaccineer recruited antibodies similar to Sanofi’s Fluzone, a higher dose, targeted at adults. Moderna shares fell 5.6 percent on the day they announced, while Sanofi rose 3 percent.
Thomas Triomphe, director of research and development at Sanofi vaccine business, said the results reflected the company’s ever-present message: “the flu is not Covid” – that the flu vaccine should be easy to distribute and have minimal side effects.
“Most importantly is the modern generation of mRNA. . . it is a major epidemic but it does not fit into the epidemic market, ”he said.

Jo Walton, a specialist at Credit Suisse, said there was a “greater risk factor for flu mRNA deficiency” than Covid, in part because flu-like death often results from cardiovascular complications and the mRNA Covid vaccine is linked to a deficient heart. results.
“The last thing you want to do is have an explanation for any heart problems in the aftermath, then the safety assessment will be great,” he said.
Sanofi’s main flu vaccine has been shown to protect against serious complications of the flu, including heart disease. The mRNA vaccine cannot guarantee this extra benefit – or it may take some time to collect the same.
Ethan Settembre, vice president of research and development at Seqirus, said it was “early days” in our understanding of how mRNA can help the flu and improve the “good” vaccines available.
“We are always trying to make a good flu vaccine,” he said. “And new technologies sometimes offer this opportunity. Sometimes they make mistakes, sometimes they don’t.
Although work has already been done to make the Covid mRNA vaccine easier to store and carry, they also need to be more careful than the flu vaccine, which is often offered in pharmacies or doctors’ offices because they do not require cold storage.
Seqirus is active in promoting its protein-based vaccine, which grows in cells, not eggs, and uses adjuvant to boost the immune system. He added that this could be a stabilizing opportunity and is proven to support strong responses from those most at risk of influenza, children and the elderly.
But it is also developing a mRNA vaccine, using a self-replicating technology that amplifies its effects, which are set to begin testing in humans by the second half of next year.
Sanofi has so far initiated a phase 1 trial of the mRNA flu vaccine. Triomphe believes the company is well on its way to success: it bought the original MRNA Translate Bio for $ 3bn earlier this year, and promised to farm. € 400m per year in mRNA vaccine research with 400 active scientists.
The French company hopes that this, along with its relationship with consumer organizations and government agencies that provide guidance on flu vaccines, will help address the risks of Pfizer, BioNTech and Moderna.
Walton of Credit Suisse believes that while the market is changing inevitably, it will take a lot to change the benefits of accountability. “If Sanofi is three years late, the worst case scenario, one year delay after mRNA vaccine manufacturers, is it enough to kill their license or not? I doubt it is not.”
[ad_2]
Source link